After the 4 f subshell is filled, the 5 d subshell is populated, producing the third row of the transition metals. 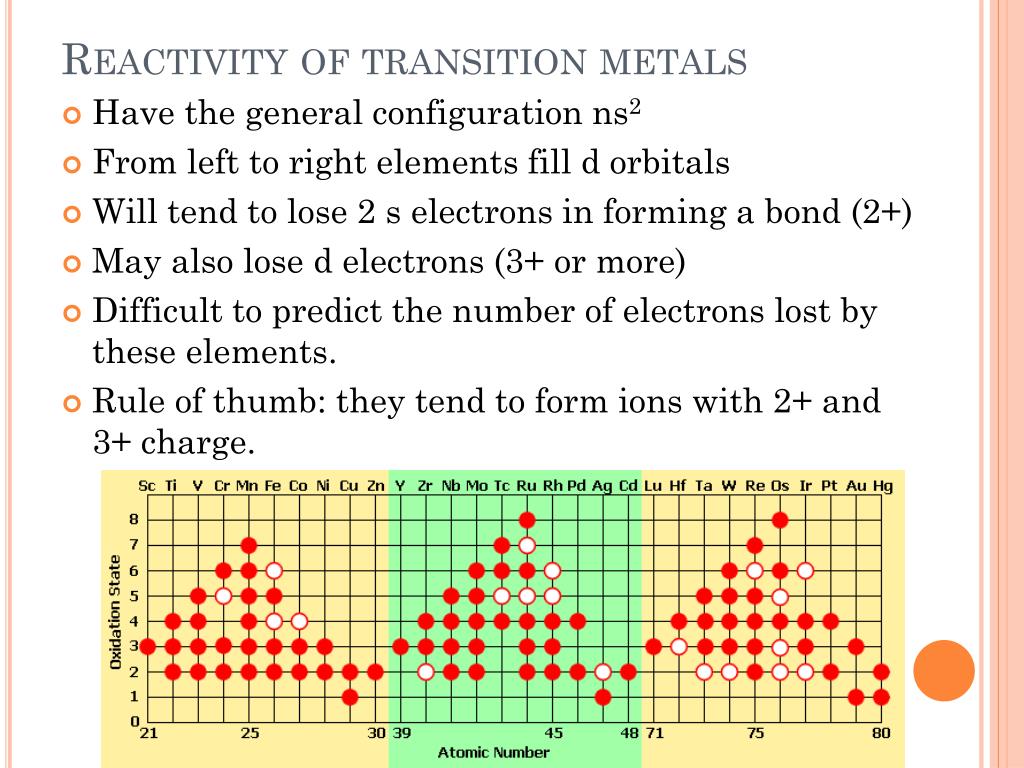
From this point through element 71, added electrons enter the 4 f subshell, giving rise to the 14 elements known as the lanthanides. Although La has a 6 s 25 d 1 valence electron configuration, the valence electron configuration of the next element-Ce-is 6 s 25 d 04 f 2. Further complications occur among the third-row transition metals, in which the 4 f, 5 d, and 6 s orbitals are extremely close in energy. For example, Nb and Tc, with atomic numbers 41 and 43, both have a half-filled 5 s subshell, with 5 s 14 d 4 and 5 s 14 d 6 valence electron configurations, respectively. In the second-row transition metals, electron–electron repulsions within the 4 d subshell cause additional irregularities in electron configurations that are not easily predicted. Table 23.1 Valence Electron Configurations of the First-Row Transition Metals Sc Because the ns and ( n − 1) d subshells in these elements are similar in energy, even relatively small effects are enough to produce apparently anomalous electron configurations. In Chapter 7 "The Periodic Table and Periodic Trends", we attributed these anomalies to the extra stability associated with half-filled subshells. Unexpectedly, however, chromium has a 4 s 13 d 5 electron configuration rather than the 4 s 23 d 4 configuration predicted by the aufbau principle, and copper is 4 s 13 d 10 rather than 4 s 23 d 9. With two important exceptions, the 3 d subshell is filled as expected based on the aufbau principle and Hund’s rule. As we go across the row from left to right, electrons are added to the 3 d subshell to neutralize the increase in the positive charge of the nucleus as the atomic number increases. 
The valence electron configurations of the first-row transition metals are given in Table 23.1 "Valence Electron Configurations of the First-Row Transition Metals". Alkali metals make an alkaline solution when reacted with water, transition metals do not.Electronic Structure and Reactivity of the Transition Metals.Transition metals have much higher melting and boiling points than alkali metals.Transition metals are much denser and stronger than alkali metals.Transition metals are not as _reactive as alkali metals _(group 1 metals).The main differences between transition metals and alkali metalsĮven though transition metals and alkali metals are all metals, there are still quite a few differences: Note: transition metals and their compounds all make good catalysts! Transition Metals vs Alkali Metals Good conductors of heat and electricity.They have the properties of metals that you would expect: The transition metals are the big bulk of elements in the middle of the periodic table (coloured in pink on the picture above). The general properties of transition metals. Which groups the transition metals occupy The larger the molecules, the more intermolecular forces (see IntermolecularForces section) Transition Elements Since they don’t react much they are also non-flammable.īoiling points increase down the group - this is because the atomic radii increases down the group. 
The noble gases have a full outer shell of electrons, therefore they don’t tend to react much (inert). This is because all of the element are gaseous at room temperature. Group 0 elements are known as the noble gases. Group 0 and Transition Elements Group0Trends
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
